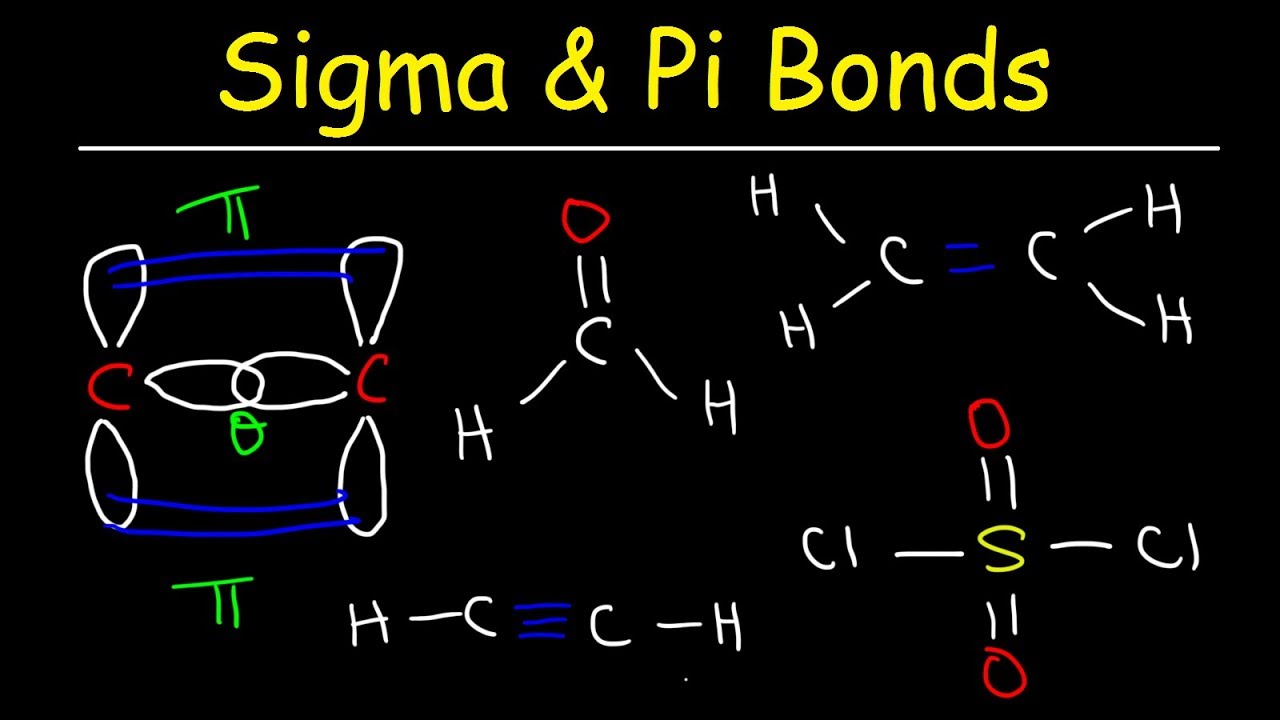
(iii) The hybrid orbitals are more effective in forming stable bonds than the pure atomic orbitals. (ii) The hybridized orbitals are always equivalent in energy and shape. (i) The number of hybridized orbitals formed is equal to the number of orbitals that get hybridized. The important characteristics of hybridisation are listed below: Thus, to form four equivalent bonds, one 2s and three 2p-orbitals of carbon hybridize and form four new orbitals.such orbitals are called sp 3 hybrid orbitals. The new orbitals formed as a result of hybridization are called hybrid or hybridized orbitals. Hybridisation may be defined as the phenomenon of intermixing of the orbitals of slightly different energies so as to redistribute their energies and to give new set of orbitals of equivalent energy and shape. The equivalent character of the bonds can be explained with the help of hybridisation. But actually, in most of the carbon compounds, such as methane (CH 4 ), carbon tetrachloride (CCl 4 ) etc., all the four bonds of carbon atom are equivalent. Therefore, all the bonds will not be equivalent. For e.g., in the formation of CH 4 molecule, one C-H bond will be formed by the overlapping of 2s-orbital of C and 1s-orbital of H whereas the other three C-H bonds will be formed by the overlapping of 2p-orbitals of C and 1s-orbital of H. The above configuration reveals that all the four bonds formed by carbon will not be identical. 
This state is known as excited state and the configuration of carbon in the excited state is: Thus, four half-filled orbitals form in the valence shell this accounts for the bonding capacity of four carbon atoms. To explain the tetravalency of carbon, it was proposed that one of the electrons from 2s filled orbital is promoted to the 2p empty orbital (2p z ), which is in a higher energy state. For e.g., in case of carbon, the ground state electronic The structures of different molecules can be explained on the basis of hybridization.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
